-
-
December 26, 2022 at 1:13 pm
Rahmani Zakaria
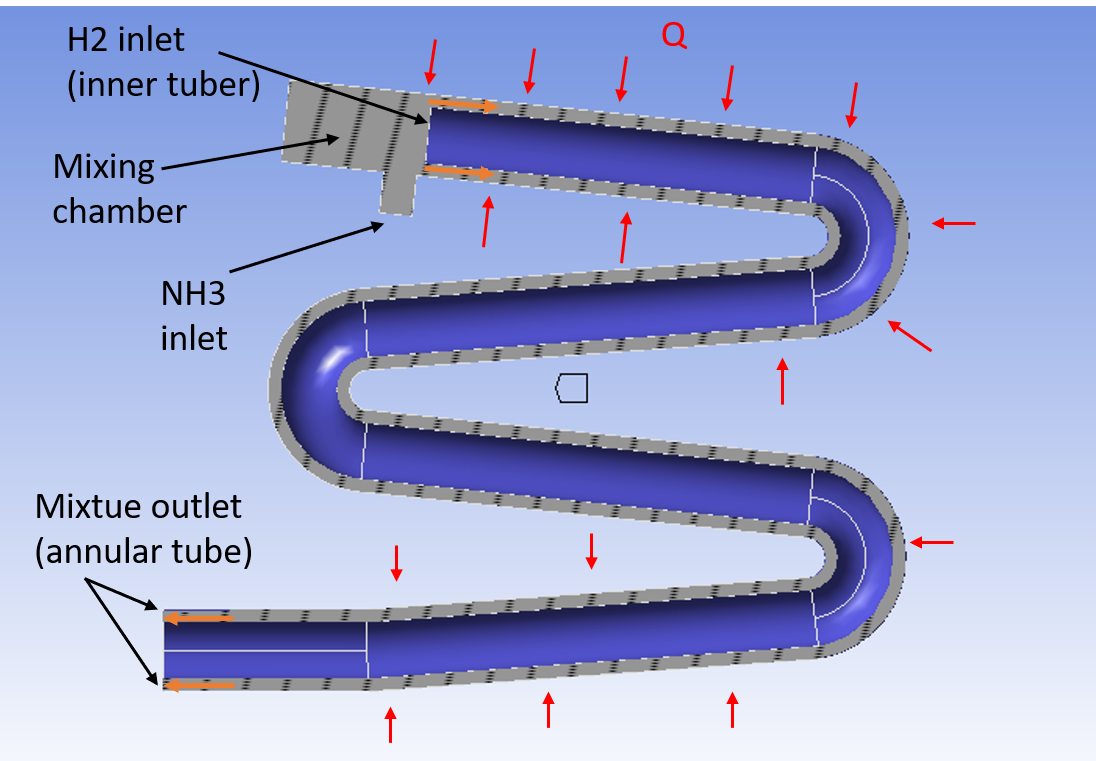
SubscriberHi everyone, I am trying to simulate the evaporation of Ammonia (Nh3). A tube-in-tube evaporator was designed for this simulation with 2 inlets and an outlet, where ammonia liquid enters the expansion chamber and mixing with hydrogen. I choose the mixture model with 3 phases as multiphase model upon the details providing by Ansys theory guide.
Primary phase: Hydrogen
Secondary phase: Ammonia-Liquid
Secondary phase: Ammonia-Vapor
Tsat of ammonia @1bar = -33C
P_total = 25bar
Boundary conditions:
Nh3 inlet: Velocity inelt 1m/s, T= -33C
H2 inlet: Velocity inlet 5m/s, T=-5C
mixture outlet: Outflow
Wall: Convection: 20 W/m^2.K, T=10C
Time step size: t=0.0001s
Number of time steps: 250
Max iterations/time step: 10
My questions:
- where should I add the total pressure of the mixture: the operating pressure or the gauge pressure?
- the total pressure is P_tot = 25bar, when ammonia become in contact with hydrogen it is supposed to reduce its partial pressure to P_Nh3 = 1bar and T= -33 C and allow ammonia to evaporate according to the Dalton's law.
the question Ansys will do that?
- I have also a problem with the convergence, there is a fluctuation in residuals then the solution diverges. Then I change the time from transient to steady, the fluctuations do not appear but the residuals still constant and the solution diverge after 60 iterations.
thanks in advance. -
December 28, 2022 at 1:59 pm
SRP
Ansys EmployeeHi,
1) Enter the value for total pressure in the Gauge Total Pressure field in the Pressure Inlet dialog box.
2) I suggest to patch hydrogen in the region during initialization and re run the simulation.
Can you please provide more details which include:
1) How you model the phase interaction and heat, mass,reaction in the multiphase panel?
2) Please provide the contours of hydrogen volume fraction and ammonia liquid volume fraction at t=0.
Thank you.
-
December 31, 2022 at 5:37 pm
Rahmani Zakaria
Subscriberthank you SRP for your suggestions, in regards to ''patch'' already i do that. The main problem is:
Ammonia liquid enter the evaporator at T=25C and P=25bar, the presence of Hydrogen (inert gas) there is to allows ammonia to evaporate. Whene Ammonia become in contact with Hydrogen, supposed its partial pressure and temperature drops to P=1bar and T=-33C (saturation temperature) respectively and the evaporation begin. The question how to do that in Ansys Fluent With these BC.
-
- The topic ‘Evaporation of Ammonia’ is closed to new replies.



-
5834
-
1906
-
1420
-
1305
-
1021

© 2026 Copyright ANSYS, Inc. All rights reserved.