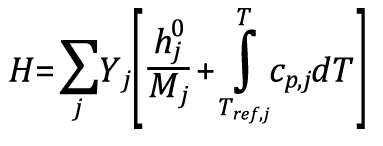TAGGED: combustion, eddy-dissipation-model, enthalpy, fluent, species-transport
-
-
February 24, 2026 at 7:53 pm
jdch232
SubscriberHello,
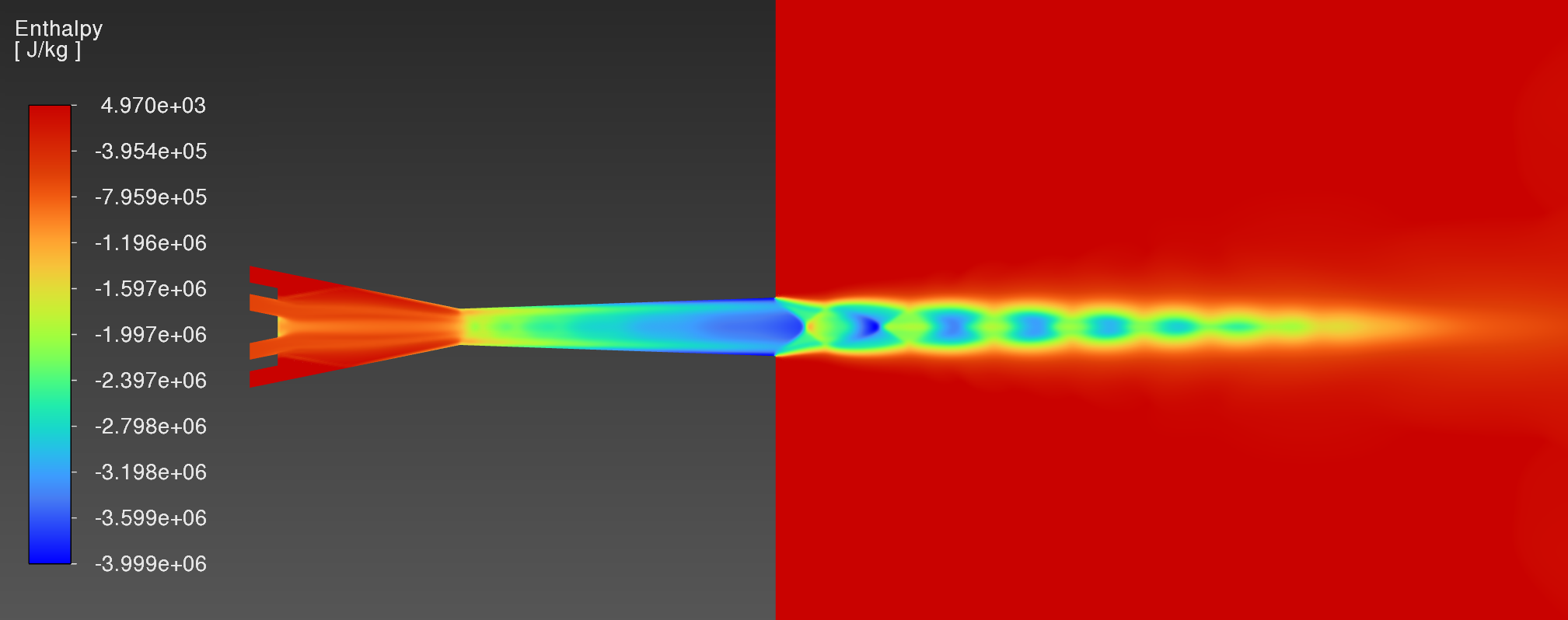
I am using Ansys Fluent to simulate a high velocity oxygen fuel (HVOF) flame impingement case. It uses density-based solver, species transport, eddy-dissipation model, and tracks propane-oxygen combustion using C3H8 + 5O2 --> 3CO2 + 4H2O. I need to find a positive recovery enthalpy against the flat plate, but I am getting negative static enthalpies in the flame, as seen in the contour below.
From the User's Manual, I know Fluent uses the equation below to calculate the mixture static enthalpy by summing across all species j. The mixture static enthalpy is negative in the flame due to the negative standard state enthalpy/heat of formation hj^0 of CO2 and H2O (-393.5 and -241.8 kJ/mol, respectively) overpowering the positive temperature-dependent specific heats. These standard state enthalpies are defined at reference temperature T_ref,j = 298.15K. Outside the flame static enthalpy is positive because only O2 and N2 are present, both of which have a standard state enthalpy of 0 kJ/mol.
Most things I've used Fluent for use the enthalpy gradient, so specific values haven't mattered, only the gradient. Now I need a positive static enthalpy to find a flame recovery enthalpy. But I am unsure how to do this.
I've been suggested not to redefine the reference temperature or standard state enthalpy as that could mess with chemistry. I think I could simply shift the enthalpy values in post-processing by adding a scalar value (would not affect the enthalpy gradient). But I am unsure of this and how much I should shift it by. Would anyone have any suggestions? Thank you.
-
February 25, 2026 at 2:10 pm
Ren
Ansys EmployeeHello,
I don't understand what you mean by "flame recovery enthalpy" but maybe you want to look at the "Sensible Enthalpy" under the Temperature category.
-
- You must be logged in to reply to this topic.



-
5199
-
1859
-
1387
-
1250
-
1021

© 2026 Copyright ANSYS, Inc. All rights reserved.