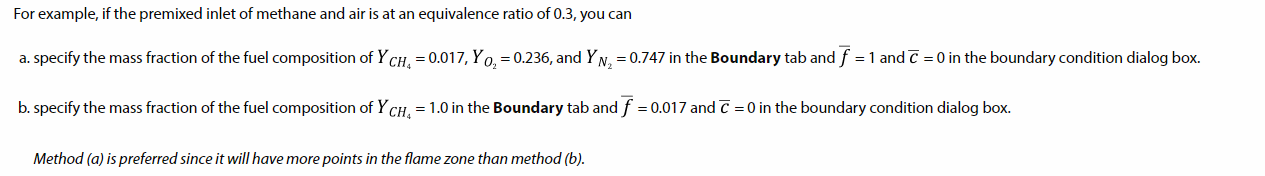TAGGED: #fluent-#ansys
-
-
August 25, 2022 at 6:19 pm
-
August 29, 2022 at 7:31 am
SVV
Ansys EmployeeHi,
May I know how you are calculating the value of mass fraction of fuel.
You can calculate the mass fraction in terms of equivalence ratio, number of moles and molar mass.
-
August 29, 2022 at 3:02 pm
britnelt
SubscriberI calculate it in terms of moles. From previous examples, I calculate N2 and O2 with a 0.21 and 0.79 ratio. So O2/(O2+N2)=0.21 and N2/(O2+N2)=0.79. Then with stiochiometric conditions, I have a ratio of 9.52:1 for Air to Fuel based on the general equation. With the equivalence ratio given, the only unknonwn in the equation is the Fuel_actual.
Equivalence Ratio=Z/(x+y) * 9.51/1 where z is the fuel_actual, x is the n2_actual and y is the o2_actual. I have 3 equations (top 2 mentioned in previous paragraphs with n2 and o2 and the third is equivalence ratio eqn) and 3 unknowns. I also know that z+x+y=1 bc it is expressed in mole fraction. I then solve for one variable to find the other variables. Is this approach correct?
-
August 30, 2022 at 5:42 am
SVV
Ansys EmployeeSorry that I wont be able to debug the math here. But in general you cannot solve mass of species in terms of number of moles alone. You need to consider the Molar mass as well. Please revisit any combustion books for solving the same. The value of mass fraction of Methane will come ~ 0.017214.
-
- The topic ‘Help on understanding Fluent Manual for Partially Premixed Combustion’ is closed to new replies.



-
5879
-
1906
-
1420
-
1306
-
1021

© 2026 Copyright ANSYS, Inc. All rights reserved.